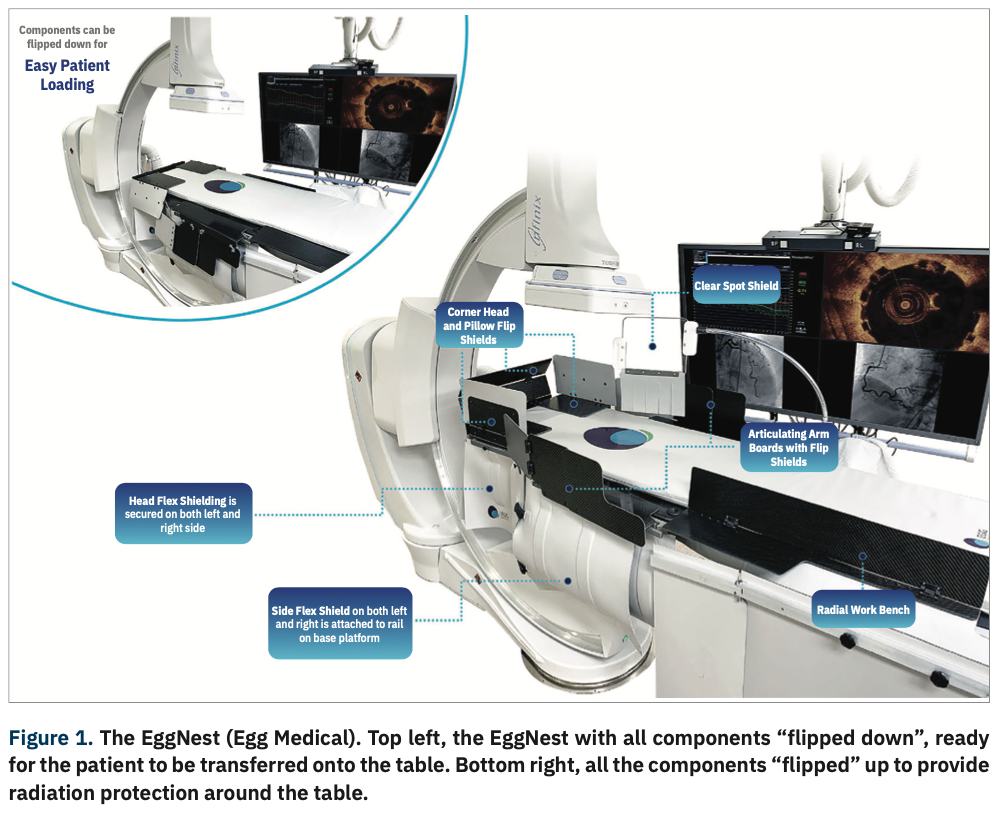
Egg Medical has secured $13m in a financing round led by TVM Capital Life Science to accelerate the commercialisation of EggNest, a scatter radiation protection platform.
EggNest is integrated into the Cath Lab’s workflow and helps to reduce up to 91% of scatter radiation exposure.
Physicians and staff who work in interventional fluoroscopy procedures are exposed to scatter radiation during medical procedures, such as stenting, angioplasty and transcatheter valve treatment.
This puts them at increased risk of long-term health effects.
The EggNest platform provides protection for staff in hospital x-ray labs, such as electrophysiology, operating rooms, cardiac catheterisation and interventional radiology, from x-ray exposure.
By dramatically reducing radiation exposure, the device protects the entire medical team regardless of location.
It swings with the x-ray, moves with the patient and provides passive protection with an integrated antimicrobial and impermeable surface.
Egg Medical CEO Robert Wilson said: “Occupational exposure of hospital staff to x-rays during medical procedures is an important workplace risk.
“Almost everyone in the interventional cardiology and radiology profession knows someone with a radiation-related illness.
“There have been minimal improvements in x-ray shielding over the past 30 years, leaving the medical teams working in these environments exposed to scatter radiation every day.”
Additionally, the company plans to utilise the funds for the development of future platforms, which will help to expand its offerings and address the unmet requirements for healthcare professionals who are at risk of radiation-related illnesses caused by long-term scatter radiation exposure.
TVM Capital Life Science managing partner Dr Luc Marengère Marengère and general partner Dr Sascha Berger will join the Egg Medical Board of Directors.
Source: MedicalDevice-Network
-
In accordance with Medical Device Authority (MDA Malaysia Ministry of Health, Malaysia), MyMedicNews serves as the leading online media in the medical device industry for Malaysia and the ASEAN region. We provide a creative one-stop web portal to deliver the latest news and product development in the medical device industry. Through our portal, key industry players can deliver business information, product, and services effectively to relevant target audience.
Disclaimer: All content is for informational or educational purposes only, and does not substitute professional medical advice or consultations with healthcare professionals. Medical device advertisements and their content are intended for Healthcare Professionals only. More information related to Malaysia medical device news, products, registration, and regulations is available on Medical Device Authority.